|

|
||
|---|---|---|---|
|
|
|||
Inertial Microfluidics for
Biophysics and Biotechnology
Welcome to Hur Research Group
Our lab focuses on developing microfluidic platforms to understand complex fluid dynamics principles and to translate acquired knowledge into practical applications. Such platforms can facilitate simple cellular assays elucidating underlying veiled relationships between cellular functions and their physical phenotypes. We utilize a unique microscale hydrodynamic phenomenon called Inertial Focusing to accomplish high-throughput target cell detection, cost-effective cell separation, and sequential multimolecular delivery. By fine-tuning microchannel geometries and flow conditions, positions of flowing microscale particles and cells can be manipulated solely by fluid forces based on their size, shape, and softness. We envision that Inertial Microfluidics will enable simple and cost effective biological assays with potential for clinical adoption to improve diagnosis and disease management quality.
Inertial Microfluidics
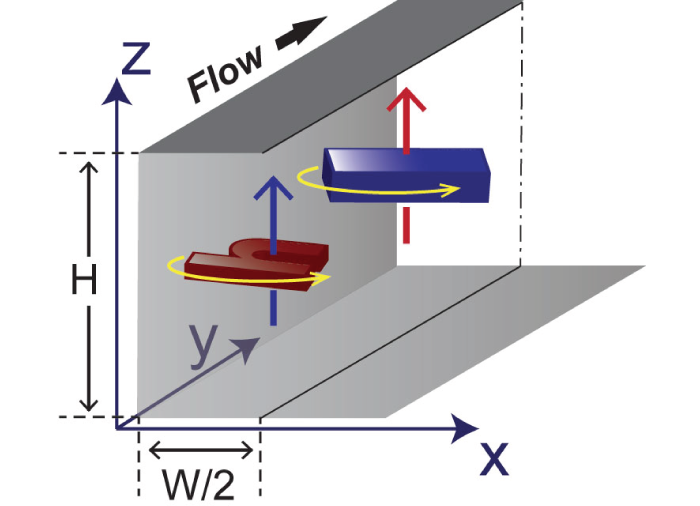
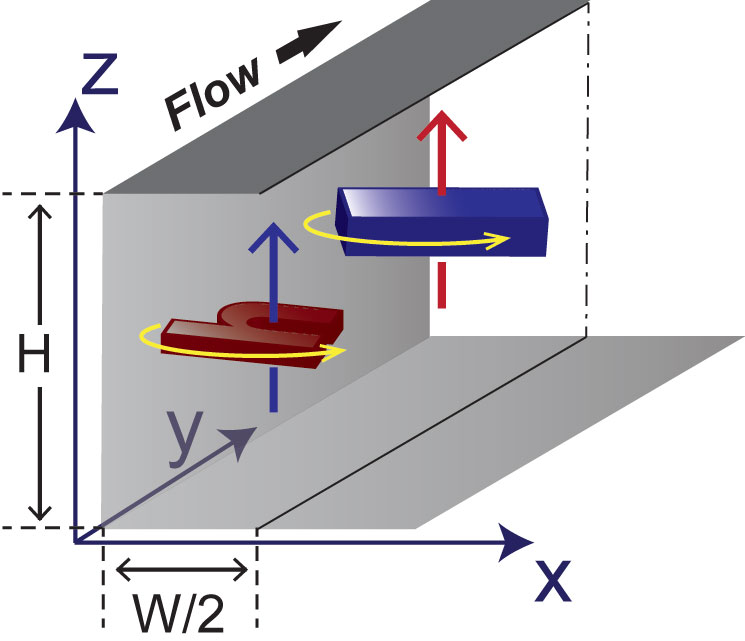
It is a common, widely-spread berief in the microfluidic community that inertia effect of the fluid can be safely neglected since the length scale of system is small, thus, the Reynolds number of the system is small. In microfluidic systems, therefore, flowing particles are believed to faithfully follow their intial streamlines. However, recently Di Carlo et al. discovered that particles can migrate across the streamlines and be focused at distinct lateral position across the channel crossection when the system is operated at relatively high flow rate, greatly resembling the inertial effect described in centimeter-scale systems in the early 1960s. Since then, many potential applications utilizing the microscale inertial effect have been demonstrated (read Inertial Microfluidics review paper for more details). In breif, a balance of counteracting inertial lift forces (specailly wall-effect and shear-gradient lift) acting on flowing particles/cells leads to unique lateral and vertical positions in a microchannel with a rectangular cross-section when flow speeds are relatively high (a few centimeter to meter per second). Since flowing particles/cells are focused to predictable locations with a uniform downstream velocity, inertial focusing holds great promises for research, clinical and industrial applications, which require precise particle focusing and manipulation in flow. Top ↑
Precise Particle Manipulation
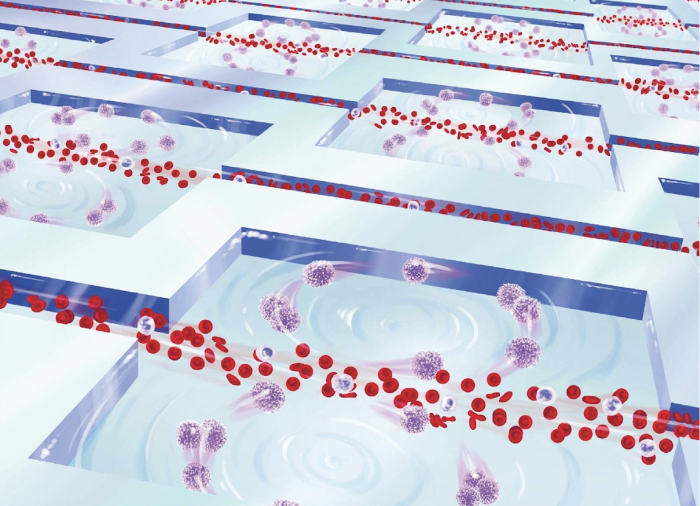
[Sheathless Flow Cytometry] Flow cytometry is the gold standard in cell analysis and it is regularly used for blood analysis (i.e., complete blood counts). Flow cytometry, however, lacks sufficient throughput to analyze rare cells in blood or other dilute solutions in a reasonable time period because it is an inherently serial process. We exploited inertial effects for label- and sheath-free parallel flow cytometry with extreme throughput (1 million cells/s) for rapid and accurate differentiation of cells. As no additional external forces are required to create ordered streams of cells, this approach has the potential for future applications in cost-effective hematology or rare cell analysis platforms with extreme throughput capabilities when integrated with suitable large field-of view imaging or interrogation methods. [Read more] Top ↑
Particles were fabricated by BiNEL.
Cellular Biophysical Property Measurement
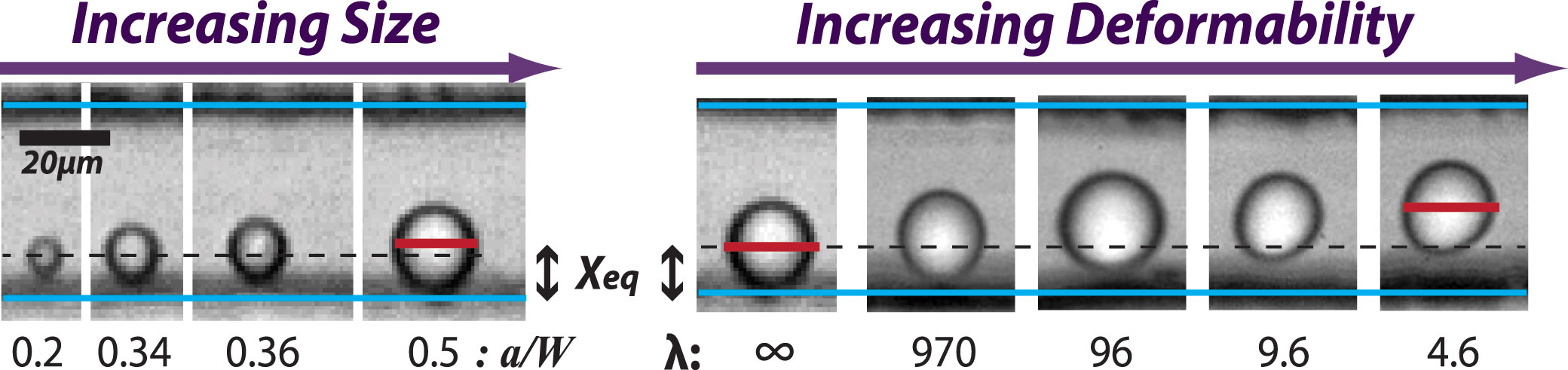
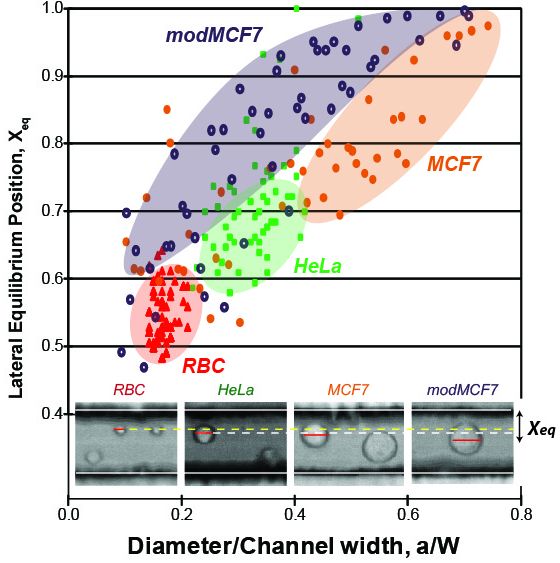
a/W: particle diameter to channel width ratio, λ: viscosity ratio between continuous and disperse phases
The fact that deformable particles experience an additional lift force suggests the possibility of high throughput deformability-induced particle classification and separation. Deformation-induced lift forces will act in superposition with inertial lift forces to create modified lateral equilibrium positions that are dependent on particle deformability.
[Phenotype Dependent Inertial Focusing] Single-cell deformability has recently been recognized as a unique label-free biomarker for cell phenotype and lineage determinations. As the flowing particles with different mechanial properties experience different magnitude of lift forces, the lateral equilibrium position can then be used as the measure of particle deformability when the particle size is taken into account. [Read more] Top ↑
Label-free Target Cell Purification

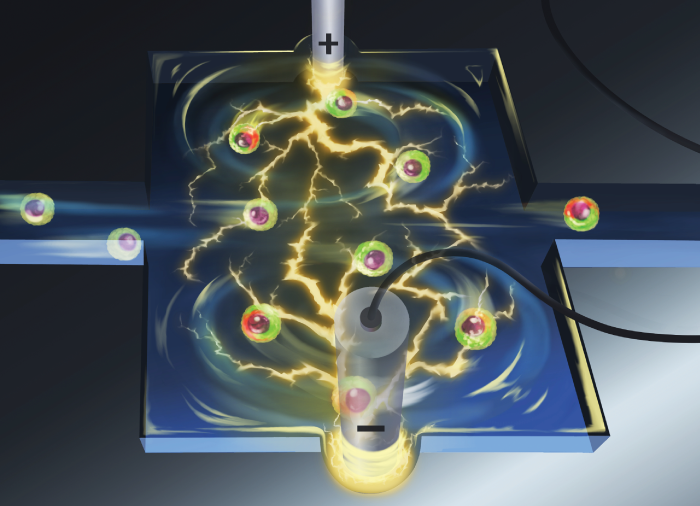
Vortex Assisted Electroporation
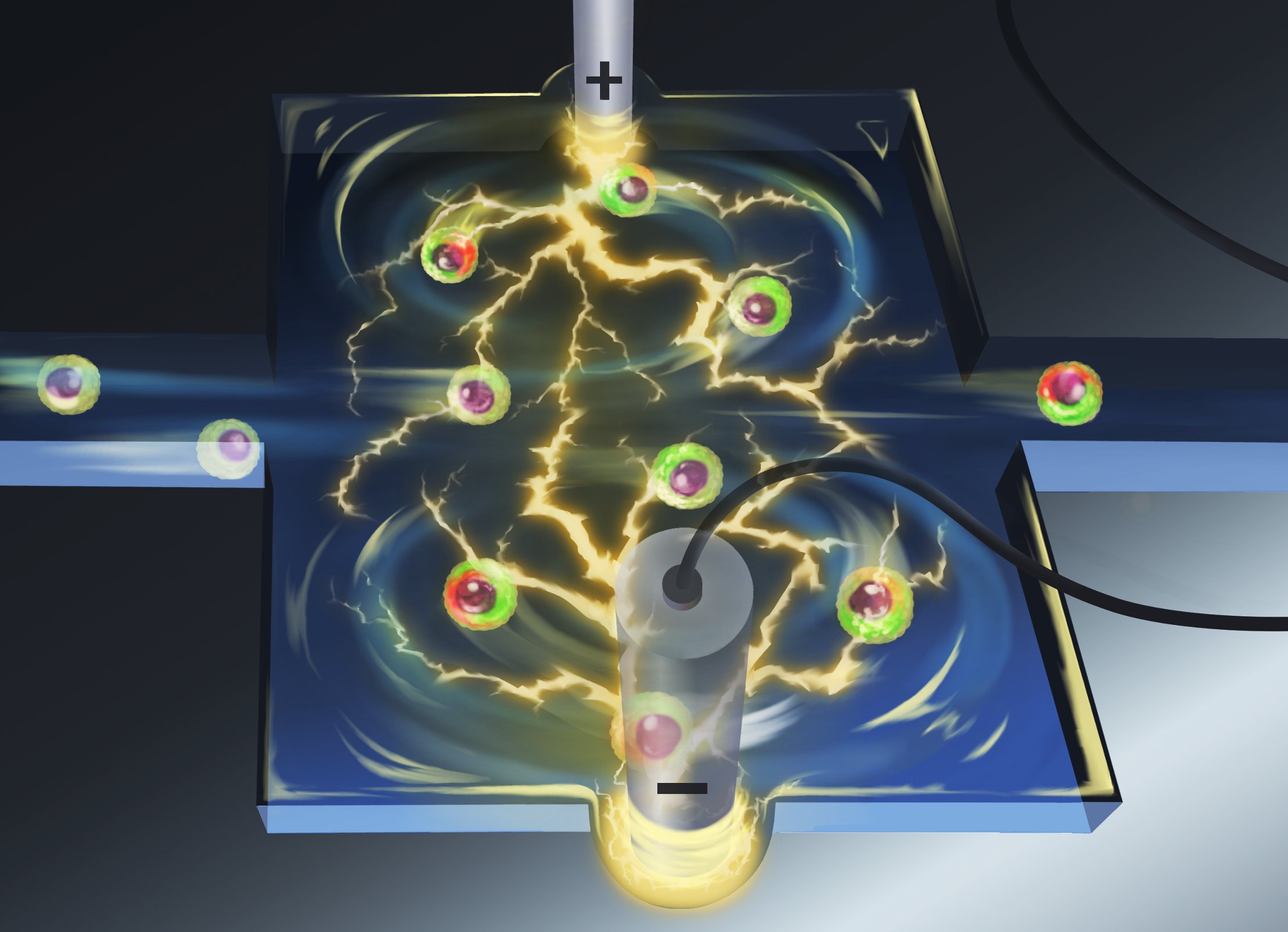
Illustration by Marc Lim .
News @fromHurLab Read more...